hermes novo nordisk | HERMES: Effects Of Ziltivekimab Versus Placebo On Morbidity hermes novo nordisk The HERMES trial is an international, multicentre, parallel group, randomized, double-blind, study in patients with HFpEF and HFmrEF, evaluating the effect of ziltivekimab 15mg compared with . 54. 7K views 5 years ago. Air Malta goes for Quality, Choice and Freshness in New In-flight Catering Chief Commercial Officer Paul Sies and Sky Gourmet Executive .
0 · IL
1 · Home
2 · HERMES: Effects Of Ziltivekimab Versus Placebo On Morbidity
Get the latest updates on Malta International Airport's arrivals and departures. Check your flight status, any delays and cancellations all in one place.
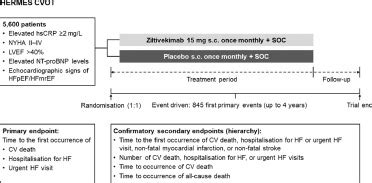
The HERMES trial is an international, multicentre, parallel group, randomized, double-blind, study in patients with HFpEF and HFmrEF, evaluating the effect of ziltivekimab 15mg compared with .

We would like to show you a description here but the site won’t allow us. Inflammation inhibition targeting the central NLRP3 inflammasome to IL-1 to IL-6 pathway of innate immunity is an emerging method for atherosclerosis treatment and .
The HERMES trial is an international, multicentre, parallel group, randomized, double-blind, study in patients with HFpEF and HFmrEF, evaluating the effect of ziltivekimab 15mg compared with placebo, given once a month, in addition to standard care, on the primary composite outcome of time to first occurrence of cardiovascular death, heart .
We would like to show you a description here but the site won’t allow us. Inflammation inhibition targeting the central NLRP3 inflammasome to IL-1 to IL-6 pathway of innate immunity is an emerging method for atherosclerosis treatment and prevention. 1 This principle was first demonstrated in the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS) using IL-1β inhibition 2 and has now been confirmed in two . IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021 May 29;397 (10289):2060-2069. doi: 10.1016/S0140-6736 (21)00520-1. Epub 2021 May 17.
In a US-based phase 2 trial, ziltivekimab, a fully human monoclonal antibody targeting the interleukin-6 ligand, significantly reduced biomarkers of inflammation compared with placebo in patients at high atherosclerotic risk. Here, we report the efficacy and safety of ziltivekimab in Japanese patients.Summary. Background IL-6 has emerged as a pivotal factor in atherothrombosis. Yet, the safety and eficacy of IL-6 inhibition among individuals at high atherosclerotic risk but without a systemic inflammatory disorder is unknown.In a US-based phase 2 trial, ziltivekimab, a fully human monoclonal antibody targeting the interleukin-6 ligand, significantly reduced biomarkers of inflammation compared with placebo in patients at high atherosclerotic risk. Here, we report the efficacy and safety of ziltivekimab in Japanese patients. Methods: RESCUE-2 was a randomized, double .
Novo Nordisk is a global healthcare company that's been making innovative medicines to help people with diabetes lead longer, healthier lives for 95 years. This heritage has given us experience and capabilities that also enable us to help people defeat other serious diseases including obesity, hemophilia and growth disorders. Abstract. Importance: The neutrophil-lymphocyte ratio (NLR) independently predicts atherosclerotic events and is a potential biomarker for residual inflammatory risk. Interleukin (IL) 1β inhibition reduces the NLR, but whether inhibition of IL-6, a cytokine downstream of IL-1, also lowers the NLR is uncertain.PLAINSBORO, N.J., May 17, 2021 /PRNewswire/ -- Novo Nordisk today presented results from RESCUE, a phase 2 randomised, double-blind, placebo controlled clinical trial assessing the effect of.The HERMES trial is an international, multicentre, parallel group, randomized, double-blind, study in patients with HFpEF and HFmrEF, evaluating the effect of ziltivekimab 15mg compared with placebo, given once a month, in addition to standard care, on the primary composite outcome of time to first occurrence of cardiovascular death, heart .
We would like to show you a description here but the site won’t allow us. Inflammation inhibition targeting the central NLRP3 inflammasome to IL-1 to IL-6 pathway of innate immunity is an emerging method for atherosclerosis treatment and prevention. 1 This principle was first demonstrated in the Canakinumab Anti-Inflammatory Thrombosis Outcomes Study (CANTOS) using IL-1β inhibition 2 and has now been confirmed in two .
IL
IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet. 2021 May 29;397 (10289):2060-2069. doi: 10.1016/S0140-6736 (21)00520-1. Epub 2021 May 17. In a US-based phase 2 trial, ziltivekimab, a fully human monoclonal antibody targeting the interleukin-6 ligand, significantly reduced biomarkers of inflammation compared with placebo in patients at high atherosclerotic risk. Here, we report the efficacy and safety of ziltivekimab in Japanese patients.Summary. Background IL-6 has emerged as a pivotal factor in atherothrombosis. Yet, the safety and eficacy of IL-6 inhibition among individuals at high atherosclerotic risk but without a systemic inflammatory disorder is unknown.In a US-based phase 2 trial, ziltivekimab, a fully human monoclonal antibody targeting the interleukin-6 ligand, significantly reduced biomarkers of inflammation compared with placebo in patients at high atherosclerotic risk. Here, we report the efficacy and safety of ziltivekimab in Japanese patients. Methods: RESCUE-2 was a randomized, double .
rolex lady-datejust 1996
Novo Nordisk is a global healthcare company that's been making innovative medicines to help people with diabetes lead longer, healthier lives for 95 years. This heritage has given us experience and capabilities that also enable us to help people defeat other serious diseases including obesity, hemophilia and growth disorders. Abstract. Importance: The neutrophil-lymphocyte ratio (NLR) independently predicts atherosclerotic events and is a potential biomarker for residual inflammatory risk. Interleukin (IL) 1β inhibition reduces the NLR, but whether inhibition of IL-6, a cytokine downstream of IL-1, also lowers the NLR is uncertain.
rolex ladies datejust keeps stopping
Home

HERMES: Effects Of Ziltivekimab Versus Placebo On Morbidity
Air Malta ceased flight operations on March 30th, 2024. We extend our heartfelt thanks to our dedicated employees for their unwavering commitment and hard work over our. 50 year tenure. We also express our gratitude to all our loyal customers, travel partners, and collaborators for their support throughout the years. Since the first Boeing 720B .
hermes novo nordisk|HERMES: Effects Of Ziltivekimab Versus Placebo On Morbidity